
Extract value from wool waste: keratin and the circular economy
Spinning a yarn: explore the chemistry of wool and use it as a raw material for biobased products through simple hand-on activities.

The following was adapted from an ILL News article.
Peas please: discover how scientists are investigating pea-based ‘milk’ foams for delicious, environmentally friendly, plant-based cappuccinos!
Due to increased awareness of the dangers associated with climate change, there has been an increased demand for plant-based foods and beverages, such as cappuccino. To make these as delicious as possible, it is vital to understand the structure of plant-based foams. Researchers from Denmark and France have successfully tackled this challenge using neutron scattering.[1]
Foams have many applications in our everyday life. For example, we are all familiar with bubble baths, shaving cream, and fire extinguishers. An area where foams are particularly important is food: baked egg white foam becomes tasty meringue, cakes are decorated with whipped cream, and frothed milk is delicious on top of a cappuccino.
Foams consist of gases dispersed within a solid or liquid. The food foams featured in this research are gas/liquid foams, in which air bubbles are enclosed by a thin film of liquid.
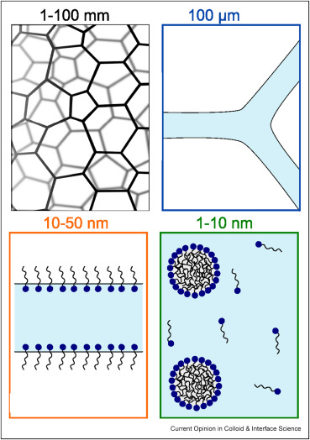
These gas/liquid foams can be unstable because the liquid around the bubbles can drain away, leading the foam to collapse. A plain water foam is very unstable, but if you zoom in further at food foams like milk foam, you see that the liquid layers are stabilized by small particles, such as proteins or micelles.
Milk foams are a widely used foam type in food technology, and researchers have studied their molecular characteristics in detail. We know that they are assemblies of air bubbles within milk, which is a mixture of water, fats, proteins, and milk sugar. When milk proteins diffuse to the air–liquid interface and concentrate there, they reduce the surface tension of the bubbles. The proteins then unfold and their hydrophilic and hydrophobic regions point towards the liquid and air phases, respectively, thereby stabilizing the foam.
In recent years, people have become increasingly aware of the negative impact that excessive consumption of animal products, including egg and milk derivatives, has on the climate. As a consequence, we are now consuming more plant-based products, including milk alternatives made with plant proteins, such as oat, soy, or pea proteins. For us to be able to enjoy plant-based cappuccinos, it is important to understand the foaming properties of plant proteins because their molecular composition and their chemical behaviour are often different from that of animal proteins. This may affect the texture, mouthfeel, and taste of plant-based foods.
A team of researchers from Denmark studies foams formed by a specific type of pea proteins known as albumins. These proteins are easy to extract from pea plants and are also very soluble, which makes them promising candidates for applications in plant-based foods and drinks, such as cappuccino. Another advantage of pea plants is that the plant grows very well at our latitudes.

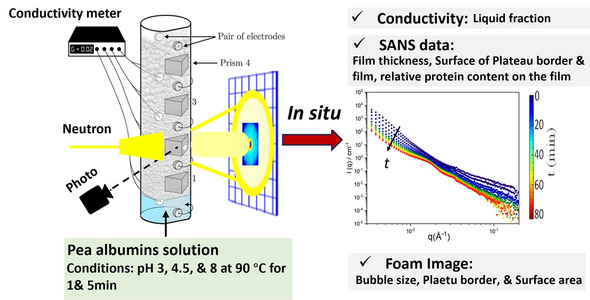
The team wanted to understand the molecular structure of pea albumin foams, how they become stabilized, and how pH and heating affect their properties. To observe these structural evolutions across a range of length scales from nanometres to millimetres, they chose a technique called small-angle neutron scattering (SANS) combined with optical imaging at the Institut Laue-Langevin (ILL) in Grenoble, France.
Neutrons are electrically neutral subatomic particles that are normally found in atomic nuclei. Free neutrons can be produced at neutron reactors such as the ILL. Using dedicated instruments, neutron beams are directed at samples such as the foams described here. The samples scatter some of the neutrons, which are recorded on a special detector. By analyzing how many neutrons are scattered at different angles, researchers can gain detailed, nanoscale information about the molecular structure and properties of the samples.
They were able to use the SANS technique to collect information on foam properties, like bubble size, film thickness, protein content, and plateau border, over time and at different pH and temperature conditions. This is important in food technology since foodstuffs can have different pH values and may be prepared or served at high temperatures. Coffee, for example, is acidic and usually served hot.
The authors studied pea albumin foams at pH values of 3, 4.5, and 8, with pH 4.5 corresponding to the isoelectric point of the pea albumin proteins. The isoelectric point is different for every protein, depending on the balance of basic/acidic amino acids it contains, and it is the pH value at which there are an equal number of positively and negatively charged amino acid residues on the protein surface. At pH values above the isoelectric point, the protein has an overall negative charge, and at pH values below the isoelectric point, the protein has an overall positive charge; in both cases, the protein molecules repel each other. However, at the isoelectric point, the protein molecules have an overall neutral charge and no longer repel each other. Instead they tend to clump together (aggregate) and can even visibly precipitate out of solution.
For the pea albumins studied here, this aggregation meant that a thick protein layer formed at the air–liquid interface of the foams at pH 4.5, thereby providing stabilization. At pH 8, the film was less thick, and at pH 3, the bubble radius was the largest and the protein film was the thinnest. These foams were particularly prone to liquid drainage (liquid diffusing through the foam), which may affect their structure.

In addition to pH, temperature is also known to affect protein structure through thermal denaturing. A familiar example is the frying of eggs, during which the initially clear and liquid egg white becomes white and solid. In the case of pea albumin foams, heating the solutions prior to foaming did not have a visible effect on foam structure. It seems that the pea albumins are less sensitive to temperature than to pH variations.
These experiments represent a very important step towards understanding the molecular properties of plant-based foods, which are becoming more and more important as sustainability becomes an increasing concern amongst consumers. It also illustrates the power of neutron scattering as a technique that allows real-time and multi-length scale characterization of highly complex biological systems.
[1] Li R et al. (2025) A time-resolved investigation at multiple-length scales of the structure of liquid foam stabilized by albumins from pea. Journal of Colloid and Interface Science 678: 1049–1060. doi: 10.1016/j.jcis.2024.09.086
[2] Chiappisi L (2024) Liquid foams: new insights and perspectives from neutron and synchrotron scattering experiments. Current Opinion in Colloid and Interface Science 72. doi: 10.1016/j.cocis.2024.101823

Spinning a yarn: explore the chemistry of wool and use it as a raw material for biobased products through simple hand-on activities.

Future food: would you bite into a test-tube burger or a Petri dish steak? How do we make lab-grown meat, and what might it mean for health, farming, and the environment?
Bacteriophages look like alien spaceships but they are actually viruses that infect bacteria. Use these fantastic beasts to explore protein stability.