Supporting materials
Download
Download this article as a PDF

You shall not pass: explore the function of deep geological repositories and the key role of bentonite in preventing the leakage of highly radioactive waste.
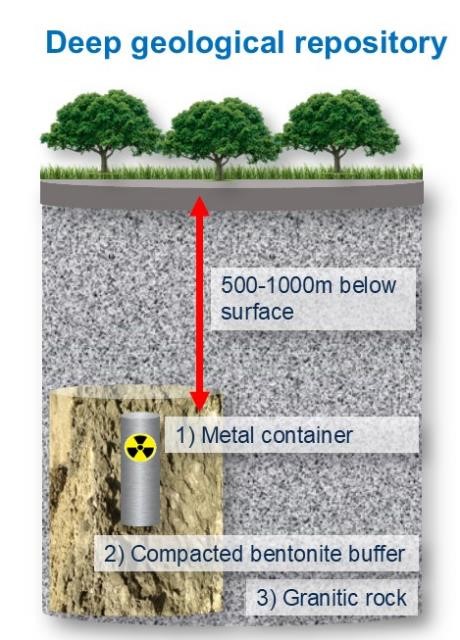
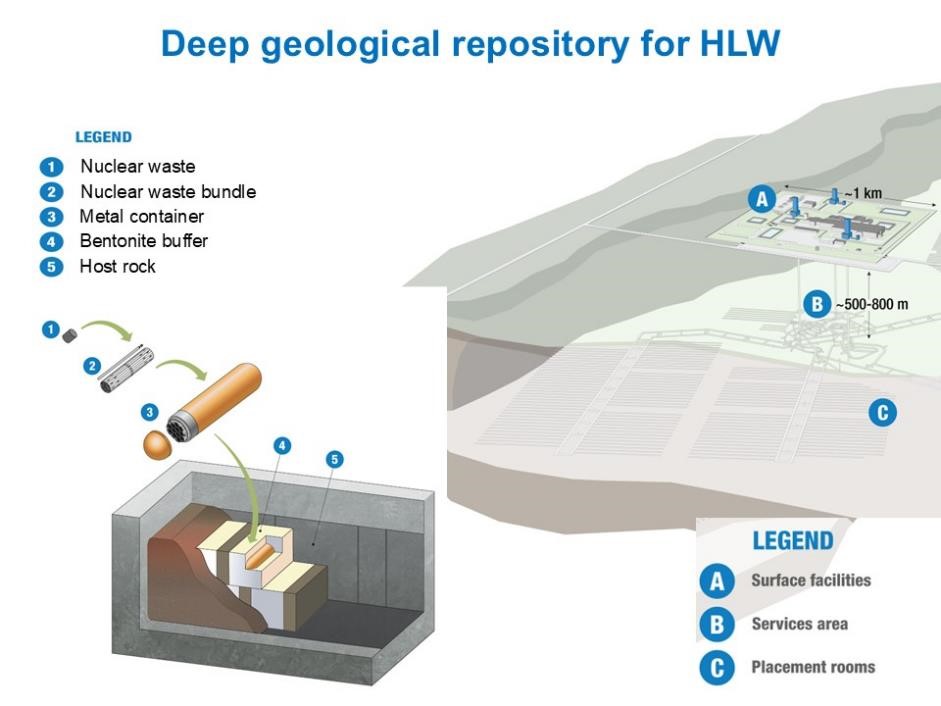
The management of high-level radioactive waste (HLW) is a serious and worldwide environmental problem. There are already tons of nuclear waste that must be safely stored for at least 100 000 years for the radiotoxicity to decrease to nondangerous levels.[1] Deep geological repositories (DGRs) are the internationally accepted multibarrier storage system for isolating highly radioactive waste. Solid nuclear waste is placed in metal containers (technical barrier) that are surrounded by a sealing and backfilling buffer material (geotechnical barrier) and buried deeply (500–1000 m underground) within a stable geological formation.[2]
Bentonite is a type of clay that can be used as a geotechnical barrier due to its good mineralogical, geochemical, mechanical, and technological properties, such as high swelling capacity and good compaction properties. These fantastic characteristics are very useful for the sealing and backfilling material to maintain the integrity and stability of DGRs of nuclear waste.
The following activities are suitable for students aged 11–16. The students will learn the importance of good isolation of nuclear waste and the great properties of bentonites in the DGRs for HLW.
In this activity, students build an interactive model of a deep geological repository (DGR). As previously mentioned, the DGR involves several barriers, for example: 1) a metal container, 2) compacted bentonite buffer, and 3) a granitic host rock. This activity consists of building a giant puzzle, where all the different pieces (barriers) must be positioned correctly for the safe storage of nuclear waste. The model can then be used as a basis to discuss the problem of managing radioactive waste and DGRs.
This activity can be performed in small working groups, where each group works on one step or part of the model. Preparation of the model will take around an hour.
Be careful when painting with the grey spray paint; if possible, do this outdoors or close to an open window.
Be extremely careful when using the box cutter (Stanley knife) to avoid cuts.
Per model:







The metal canister in which the spent nuclear fuel is encapsulated is the first barrier, a technical barrier, isolating the waste.
The geotechnical barrier, often compacted bentonite, plays an important role in the safety of a DGR due to its favourable properties, such as high swelling capacity, very low permeability, good compaction properties, and good cation-adsorption capacity. Thus, bentonites will contribute to maintaining the integrity of the metal canisters, acting as a buffer against temperature and rock movements. Also, in the worst-case scenario of accidental leakage, bentonites contain, prevent, and retard the dispersion of radionuclides into the environment.[3]
The geological barrier or host rock (the natural barrier) mainly provides mechanical stability to the DGR. It must be a low-permeability rock to prevent and retard any possible leakage from the repository system.
The following questions can be used to lead further research and discussion of the activity:
By carrying out this activity, students will learn and be aware of the importance of worldwide repository barriers by building and playing with the giant puzzle.
Compacted bentonites are used as a geotechnical barrier in nuclear waste repositories to provide good thermal, hydraulic, mechanical, and chemical isolation, to ensure the long-term stability of the DGR.
In this activity, coloured water, representing radioactive waste, is added to different bentonite types (pellet and powder) to check the great swelling capacity of bentonite and how it can block radioactive filtration to the host rock and biosphere. Depending on the bentonite compaction grade, radioactive leakage will take more or less time to go through the bentonite, thus reaching and contaminating the environment.
This activity can be performed in groups or individually. Each group/student should be assigned different water volumes.
This activity takes 20–30 minutes and the final results can be checked after some hours and the next day, for better comparison.
When manipulating bentonite powder, do it gently because there is a respiratory risk due to the small particles.


The following questions can help to discuss the observations and understand the underlying processes:
When dry, the clay particles in bentonite are tightly packed together and have a small surface area exposed to the surrounding environment. However, when hydrated, water molecules are able to enter the spaces between the clay particles, causing them to expand and create a larger surface area. This expansion causes the bentonite to swell and become gel-like in consistency.
After 30 minutes, liquid radioactive waste will be absorbed, while bentonite powder is not visually affected. In the case of bentonite pellets, they will be partially broken down. The next day, liquid radioactive waste will be fully absorbed by bentonite, which will be swollen, and in the case of pellets, their size will be greatly increased. Bentonite pellets are compacted bentonite; therefore, they can absorb a higher water volume.
This fantastic property of bentonite prevents dispersion of the radioactive waste through the DGR if the metal container becomes corroded, allowing water to seep through, and therefore, it will protect the environment and humanity.

By performing this activity, students will easily learn about, and check for themselves, the great swelling capacity of bentonite and get a better sense of how important it is for the DGRs of nuclear waste. The properties of bentonite barriers in DGRs are still an active research topic, and as an optional extension, students can read the current research infosheet to learn about my research on microbial activity at the bentonite barrier in DGRs of nuclear waste.
The author acknowledges funding received from the EU.

[1] Hedin A (1999) Deep repository for spent nuclear fuel. Technical report: TR-99-06. Swedish Nuclear Fuel and Waste Management Company.
[2] IAEA (2003) Scientific and technical basis for the geological disposal of radioactive wastes. International Atomic Energy Agency.
[3] Svensk Kärnbränslehantering AB (2010). Data report for the safety assessment SR-Site. Technical Report TR-10-52 SKB. Swedish Nuclear Fuel and Waste Management Company.
Download this article as a PDF

On a roll: a humble roll of toilet paper can be used in science experiments explore diverse topics in materials science, chemistry, and physics.

I’ve got the power: meet a simple device that can create a powerful sterilizing solution from just air, water, and electricity.

Stacking up: use common household items like coins and paper explore one of the most significant scientific discoveries of the 19th century.