
Plant solar power: unlocking the secrets of photosynthesis with X-ray free-electron lasers
Capturing the moment: to use artificial photosynthesis to produce clean energy, we need to better understand it first. Learn about how X-ray…

Invisible soap, heavy water and a research reactor come together to provide enlightening insights into how plants and bacteria turn light into food.
Photosynthesis is a remarkably intricate biophysical process in which quantum physics meets biology.[1] It allows plants, algae and certain bacteria to perform what mankind can only dream of: transforming sunlight straight into nutrients such as sugars with incredible efficiency. In light of the ongoing energy crisis, scientists have been wondering for a long time whether it is possible to design an artificial replica of photosynthesis. Such a technology, which relies purely on sunlight and water, would immediately solve one of humanity’s greatest challenges: the quest for sustainable, renewable energy production.
While artificial photosynthesis is still an unachieved goal, researchers continue to work towards a better understanding of this complex process, in the hope that this knowledge will lead to the development of novel energy technologies. To understand the future steps towards our goal of replicating photosynthesis, let us first take a look at what we already know.
The magic happens in tiny compartments known as chloroplasts.[1] If we zoom in further on these small powerhouses, we discover that they contain so-called thylakoids – stacks of lipid membranes that contain a well-organised sequence of membrane proteins. These proteins are what drives the light-dependent reactions of photosynthesis.
After light is absorbed by a first protein complex known as photosystem II, a chain reaction is triggered that initiates a fast electron transfer between several other protein complexes, including photosystem I. Simultaneously, water molecules are split into oxygen and protons.
At the end of the chain reaction, electrons and protons are used to generate chemical substances which are then further metabolised into nutrients and building blocks that the cell needs to survive.
A particular focus of scientists trying to decipher the molecular intricacies of photosynthesis has been on the proteins mentioned above. To understand how they work on a molecular level, we need to look at their overall shape and structure since in biology, these two aspects are often closely related.
Enzymes are a good example of how structure and function are often closely related in biological systems. These proteins are frequently built in a way that allows them to fit tightly around their molecular targets, thereby facilitating biochemical reactions.
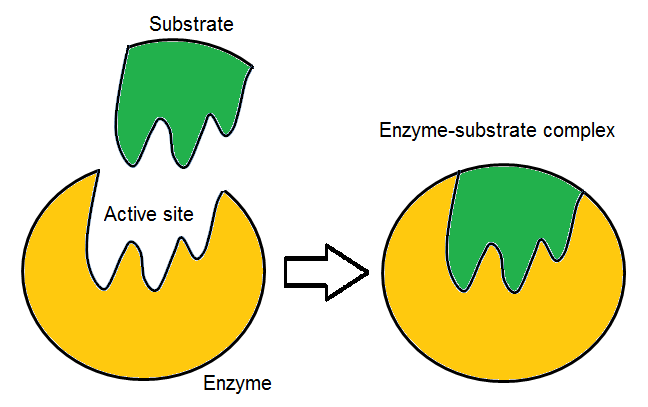
One method scientists use for such structural studies is so-called small-angle scattering, where particles are scattered off biomolecules in solution.
Neutrons are electrically neutral subatomic particles that are normally found in atomic nuclei. Free neutrons can be produced at neutron sources such as the Institut Laue-Langevin (ILL) in Grenoble, France.[2] Some of their properties enable them to yield information that would otherwise be impossible to obtain using other techniques. This makes them a powerful probe to study small samples of materials.
Using dedicated instruments (Figure 3), neutron beams are directed at samples such as photosystem I and II. Some of the neutrons are scattered by the samples, which is recorded on a special detector.
By analysing how many neutrons are scattered at different angles in a small angle neutron scattering (SANS) experiment, researchers can gain detailed nanoscale information about the molecular structure and properties of the samples.
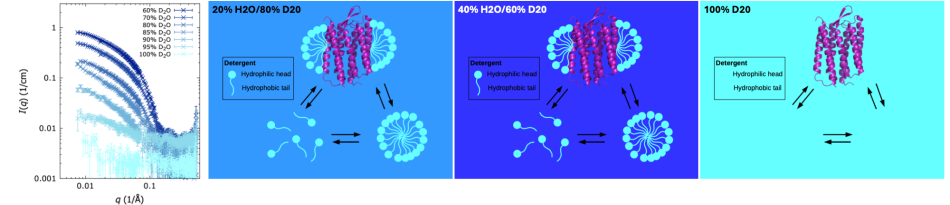
Importantly, neutrons interact more strongly with hydrogen (H) than with its isotope deuterium (D), meaning that they don’t scatter much when hitting deuterium, creating almost no signal. If some of the hydrogen in the sample is replaced with deuterium, selected parts of the sample can be hidden or highlighted for even more detailed studies.

An important challenge when studying these proteins is that they require a hydrophobic (lipid) environment to maintain their structure and function (remember that in plants, they are embedded in hydrophobic thylakoid membranes).[1] As soon as we try to take them out of their native environment and put them into aqueous solutions to run tests such as scattering experiments on them, they start malfunctioning.
Scientists therefore need to use soap-like molecules, known as detergents, to stabilise photosynthetic proteins in solution. Unfortunately, detergents come with their own challenges: they interfere with structural studies since their own molecular signature becomes visible along with that of the proteins of interests. Researchers can therefore overestimate the protein size or obtain wrong information on their shape.
In a recent study on photosystem I from the bacterium Thermosynechococcus elongatus, researchers exploited a powerful asset of small-angle neutron scattering to tackle these problems.[3] Due to the strong difference in the way neutrons interact with hydrogen and deuterium, custom-designed detergents (Figure 4), in which some of the hydrogen has been replaced by deuterium, can be made invisible in a neutron scattering experiment while keeping the fragile photosynthetic proteins intact and functional. Thanks to this ‘invisible’ detergent, the team of scientists behind the study were able to obtain a much more precise neutron-based estimate of the photosystem than in experiments where the detergent was partially visible.
In combination with other scattering techniques, such as small-angle X-ray scattering, this approach is an important step forward in our understanding of photosynthesis.
Although we are not yet able to copy nature’s masterpiece of photosynthesis, studies such as this one help us gain insight into this fascinating conversion of ‘light into life’. With each experiment, we are getting closer and closer to designing highly efficient energy solutions rooted in plant-based technology that will hopefully become reality one day.
[1] Piechulla B, Heldt HW (2015) Pflanzenbiochemie 5th edition. Springer. ISBN: 9783662654286
[2] The Institut Laue-Langevin (ILL): https://www.ill.eu/
[3] Golub M et al. (2022) “Invisible” detergents enable a reliable determination of solution structures of native photosystems by small-angle neutron scattering. The Journal of Physical Chemistry B, 126: 2824–2833. doi: 10.1021/acs.jpcb.2c01591

Capturing the moment: to use artificial photosynthesis to produce clean energy, we need to better understand it first. Learn about how X-ray…

Use thin-layer chromatography to discover the variety of pigments that play a role in photosynthesis and give leaves their…

When next teaching photosynthesis, try these simple experiments with variegated…