More than meets the eye: the electromagnetic spectrum
Claudia Mignone and Rebecca Barnes take us on a tour through the electromagnetic spectrum and introduce us to the European Space Agency’s fleet of…

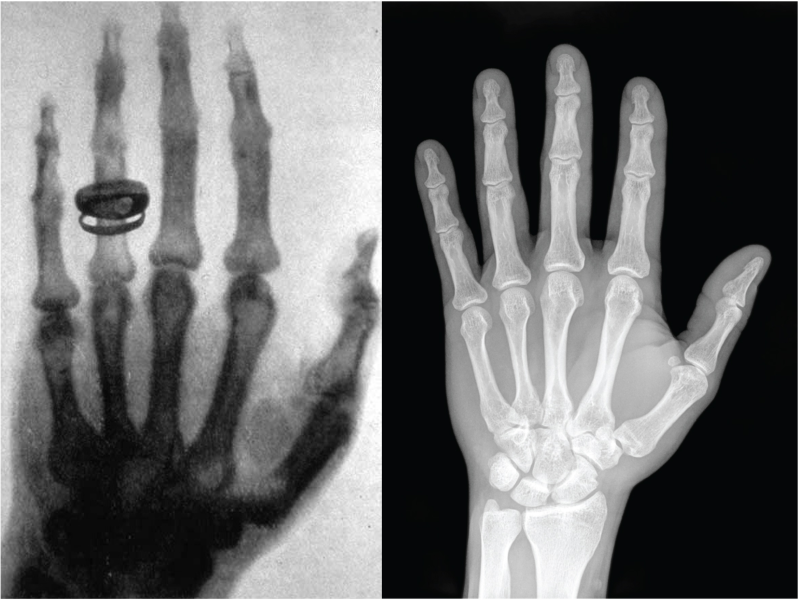
X-ray light does not only enable us to look at our bones, it also helps scientists to analyse tiny molecules that make up all living things.
In 1895, the physicist Wilhelm Conrad Röntgen noticed a strange kind of radiation that could pass through paper and flesh, but not through bone. He called it X-radiation, because he didn’t know what it was.[1,2] Today, we still call it X-ray, although in some languages they are known as Röntgen light or Röntgen rays, and they are used every day in hospitals to make the inside of the body visible.
But what if you want to make something much smaller than a broken bone visible, such as a cell, a virus, or even a single protein molecule? A normal hospital X-ray won’t do the job. In order to reveal such tiny structures, scientists need a very special kind of X-ray light: extremely intense, ultra-short X-ray flashes. Those are produced, for instance, by the European XFEL (the European X-ray Free‑Electron Laser) in Germany.[3]
It may come as a surprise to learn that X-rays are a form of light, given that we can’t see them with our naked eyes. In physics, however, the term ‘light’ means more than just what we see. Ultraviolet light, for instance, is invisible, but we notice its existence when we get sunburnt. All types of light, including radio waves, microwaves as well as X-rays, belong to the same family: electromagnetic waves.[4]
The main difference between electromagnetic waves is the wavelength. If we imagine a ray of light as a wavy line, the distance between one wave peak and the next is different for each type of light. Visible light has wavelengths around a few hundred nanometres (nm). That is an extremely short length – about 1000 times shorter that the thickness of a sheet of paper. X‑rays have wavelengths that are even shorter, down to a fraction of a nanometre.
That detail is important because the wavelength determines what you can see. For example, if you try to draw a detailed picture using a chunky marker, you can’t make very fine lines. Likewise, light with long wavelengths cannot reveal very small details. In order to make tiny structures visible, you need short-wavelength light.
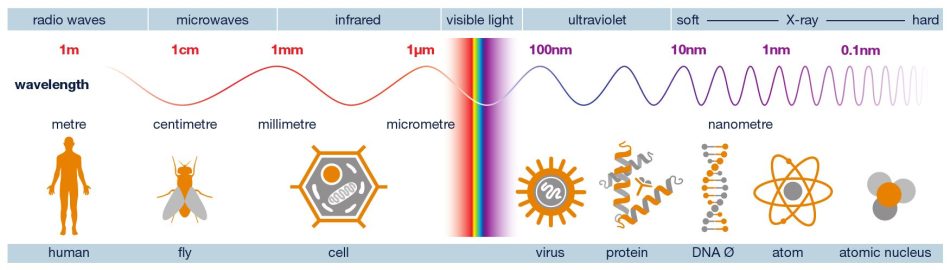
A single human cell might be only a few micrometres in size, comparable to the thickness of a sheet of paper. That’s already near the limit of what can be resolved with ordinary light microscopes. Looking at the molecular level, proteins are typically only a few nanometres in size,[5] and atoms even smaller. At those scales, the wavelengths of visible light are simply too long to pick out fine detail.
X‑rays, however, have wavelengths comparable to distances between atoms. That means they can probe matter at the atomic level, not by taking a normal photograph, but by interacting with the way atoms are arranged.
This is why X‑rays are so important in modern science. If you want to understand how a virus is built, how a protein folds into its working shape, or how a new battery material stores energy, you often need information about where the atoms are.
In a hospital, X‑ray imaging works a bit like shadow theatre. An X‑ray beam is sent through the body onto a detector. Soft tissue lets more X‑rays through, while bones absorb more. The detector records these differences, and the result is an image where bones appear bright and soft tissue appears darker.[6] The picture is created mainly by absorption, i.e. how much X‑ray intensity is lost on the way through.
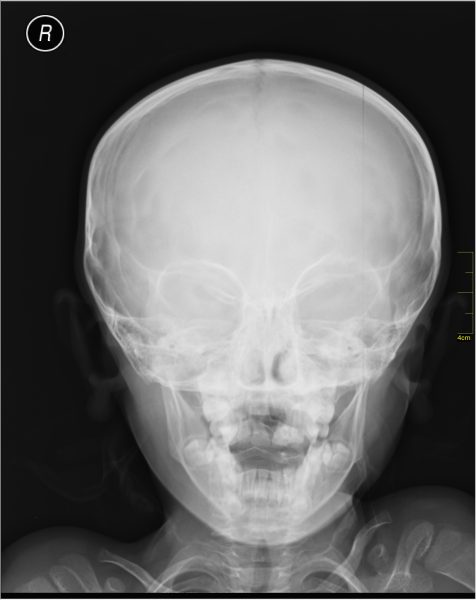
This method is quick and extremely useful in the field of medicine, but it does have its limitations. A shadow image tells you that “something blocks X‑rays here”, not exactly how atoms are arranged. To reveal tiny structures, such as the shape of a protein, you need a different strategy.
At European XFEL, scientists often don’t get a familiar picture at all. Instead, the detector records a diffraction pattern: a pattern of spots, rings, or speckles created when X‑rays scatter off a sample.[7] You wouldn’t see these effects in an X-ray of your bones, since they only appear when the scale of what is pictured is similar to the wavelength. For example, the distance between atoms in a molecule is roughly as wide as the wavelength of X-ray light.
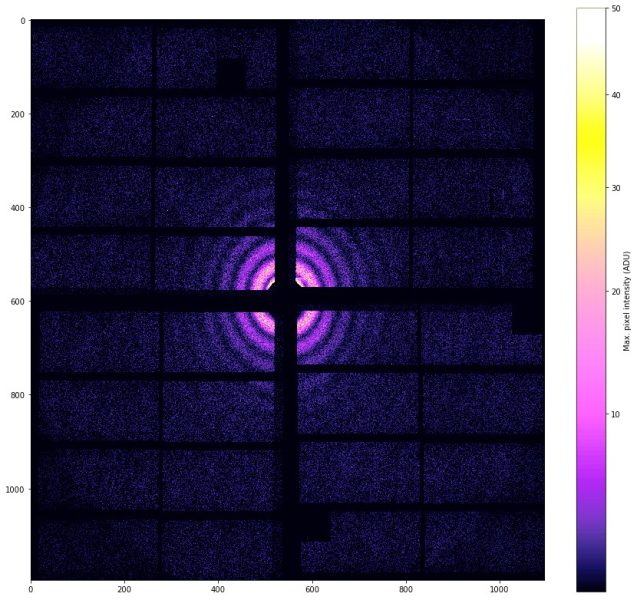
To understand diffraction, think about waves on water. If a wave passes through a narrow gap, it spreads out. If it meets an obstacle, it bends and creates patterns where waves strengthen or cancel each other. Light waves do the same. When X‑rays hit a tiny object, the waves scatter in many directions. Where these scattered waves overlap, they interfere, adding up in some places to create bright spots and cancelling out in others to create dark regions. The result is a diffraction pattern.
To the untrained eye, a diffraction pattern may resemble an abstract artwork. However, to scientists, it is a coded message about the sample’s structure. The exact spacing and brightness of the pattern depend on how atoms are arranged in the sample.
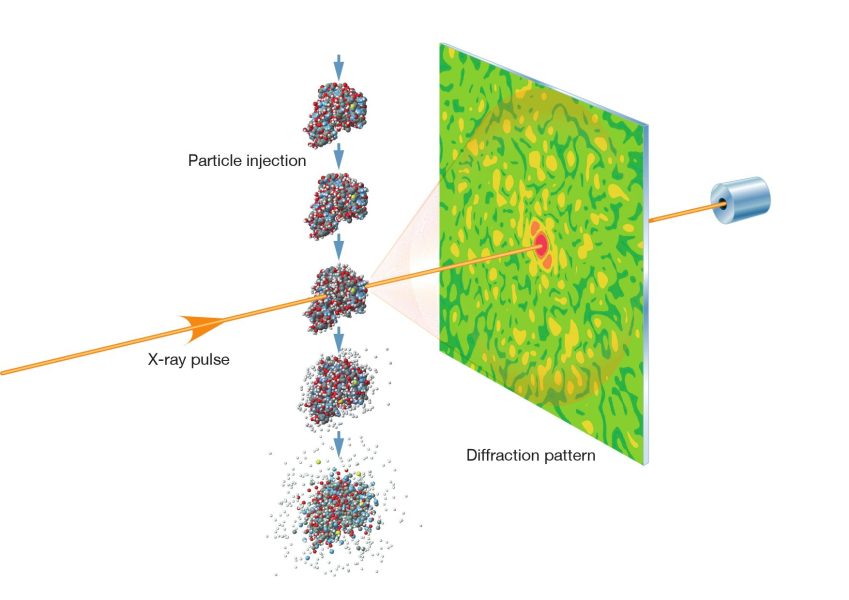
So how do you turn a pattern into a structure? The basic steps are simple to describe. First, an intense X‑ray flash illuminates a sample. The sample scatters the X‑rays, and a detector records the diffraction pattern. Then computers use mathematics and physics to work backwards and reconstruct a model of the sample’s structure.
In some experiments, scientists use crystals, which exhibit highly ordered structures.[8] Their repeating pattern of atoms produces strong, clear diffraction spots. In others, especially at X‑ray free-electron lasers, researchers study tiny crystals, or even single particles, such as viruses. In these cases, diffraction patterns are collected from many particles, often in different orientations, and then combined to build up a 3D picture.
It’s a bit like trying to identify the shape of a key from its shadow, yet more challenging: the ‘shadow’ is made by waves interfering with each other, and the object may be changing or damaged. That’s where European XFEL’s special X‑ray flashes come in.
European XFEL produces X‑ray pulses that are both extremely bright and extremely short, often only tens to hundreds of femtoseconds long (a femtosecond is a millionth of a billionth of a second). The facility can also produce a very high number of pulses per second, delivered in rapid bursts.[9]
These features matter because many samples are delicate, and many processes in nature are fast. If you want to understand how a chemical reaction happens, it’s not enough to know what the starting and finishing molecules look like. The interesting part is the in‑between steps – the brief moments when bonds break and form. As these events happen so incredibly quickly, a very short X-ray flash is needed to ‘freeze’ the motion in an image. Otherwise, the image would be blurry. Photographers use a flash for the same reason when they want to capture something that’s moving very fast.[10]
With a technique called pump-probe, a first laser pulse can initiate a reaction, and an X‑ray pulse can then ‘take a snapshot’ a tiny fraction of a second later. By changing the delay between the pulses, scientists can create a slow‑motion film of molecular change.
The brightness helps when the sample is tiny. The smaller the object, the fewer X‑rays it scatters. A very intense X‑ray pulse increases the chance of recording a useful pattern.

European XFEL is not just a bigger X‑ray machine. It is a tool for answering questions that are difficult or impossible to tackle with ordinary X‑ray sources. It helps make things visible that were invisible before.
In biology, knowing the exact structure of proteins and other biomolecules helps explain how life works at a molecular level. For instance, many medications work by fitting into a protein like a key into a lock.[11] If you can see the protein – the lock in this analogy – in detail and observe how it changes shape while it works, you can design more efficient drugs – the keys – and understand diseases more deeply.
In the fields of chemistry and energy research, observing the movement of atoms during a reaction can reveal why some catalysts speed up reactions while others do not, and how charge travels through solar cells or battery materials. In materials science, establishing the link between atomic structures and properties such as strength, conductivity, or magnetism can guide the design of new materials for technological applications.
In all these cases, European XFEL enables scientists to achieve something what seems almost impossible: using flashes of X‑ray light to ‘see’ structures far smaller than any lens-based microscope can resolve, often not as a photograph, but as a carefully decoded pattern of scattered light.

Wait! Didn’t we say X-rays are invisible? Good catch. What we are actually seeing here is glowing nitrogen molecules that have interacted with the extremely intense X-ray beam and been caused to light up as the beam travels through the air. Although the European XFEL X-ray beam is extremely intense, the glow induced in the nitrogen molecules is still relatively weak and not so easy to see with the naked eye. This image was only possible in complete darkness and by condensing 90 seconds of capture time into one photo. Like X-rays, our photographer has also managed to make the invisible visible!
Wavelength: Distance between two wave peaks. Shorter wavelengths can reveal smaller details.
Diffraction: The bending/spreading of waves and the patterns produced when waves interfere.
Diffraction pattern: The spot/ring/speckle pattern recorded when scattered waves interfere. It contains information about the structure.
Femtosecond (fs): 10–15 seconds (0.000000000000001 s)
Free‑electron laser (FEL): A type of laser that uses fast electrons to generate very intense light, including X‑rays.
[1] The discovery of X-rays: https://www.dpma.de/english/our_office/publications/milestones/inventionsthatmadehistory/roentgensx-rays/index.html
[2] Röntgen WK (1896) A New Form of Radiation. Science 3: 726-729. doi: 1126/science.3.72.726
[3] The European XFEL facility: https://www.xfel.eu/facility/overview/index_eng.html
[4] Mignone C, Barnes R (2011) More than meets the eye: the electromagnetic spectrum. Science in School 20: 51–59.
[5] The size of a protein: https://book.bionumbers.org/how-big-is-the-average-protein/
[6] A detailed overview of how X-rays work: https://my.clevelandclinic.org/health/diagnostics/21818-x-ray
[7] Fundamentals of X-ray diffraction methods: https://www.fhi.mpg.de/1075089/elena_willinger__fundamental_of_x-ray_diffraction__141107.pdf
[8] An overview of what crystal are: https://letstalkscience.ca/educational-resources/stem-explained/what-a-crystal
[9] Facts and Figures about the European XFEL: https://www.xfel.eu/facility/overview/facts_amp_figures/index_eng.html
[10] An explanation to high speed photography: https://www.diyphotography.net/high-speed-photography-setup/
[11] The lock-and-key model for drugs: https://www.biotechacademy.dk/en/identification-of-the-target-and-its-structure/

Claudia Mignone and Rebecca Barnes take us on a tour through the electromagnetic spectrum and introduce us to the European Space Agency’s fleet of…

Take a closer look at the construction of European…

Dominique Cornuéjols from the European Synchrotron Radiation Facility introduces us to the world of crystallography. It’s not all shiny…