Supporting materials
Instructions for the activity (Word document)
Instructions for the activity (PDF file)
Download
Download this article as a PDF

Around 1.5 billion people worldwide are overweight or obese. Are we just eating too much or can we blame our genes? Here’s how to investigate the genetics of obesity in the classroom.

Obesity is a big problem, in more ways than one. More than 10% of the world’s adult population is obese; in some countries this rises to 40%. Heart disease, type 2 diabetes, some cancers, pregnancy complications, joint problems, depression and anxiety have all been linked to obesityw1.

Readily available calorie-rich foods and a more sedentary lifestyle are usually blamed for the startling rise in obesity, but might our genes be part of the problem? This article introduces some of the current research into the genetics of obesity along with a method developed by the European Xplore Health project for extracting DNA, the first step for researchers looking to identify possible ‘fat genes’.
The DNA extraction would be suitable for intermediate (aged 14-16) or advanced (aged 16+) students and could be completed in one lesson. If this method is not appropriate for your students or lab, try one of two alternative methods (based on the same principles, but using more readily available materials), using frozen peas (Madden, 2006) or kiwi fruitw2. Instructions for the activityw5 can also be downloaded as Word or PDF files from the Science in School website.
The topic of obesity also provides a useful way of introducing current genetic research and ethical issues within a context that students are likely to have some personal experience of and opinions about. After all, how many teenagers don’t like fast food and lazing about in front of the TV? Other resources, including a debate pack for exploring the issues further, are available on the Xplore Health websitew3.
Someone is considered obese if they have a body mass index (BMI) of 30 or more. The healthy range is 18.5-25. BMI is calculated by dividing a person’s weight in kilograms by the square of their height in metres.
BMI = weight (kg)
(height (m))2
Although BMI provides a useful guide, it doesn’t allow for variation in build and body composition. For example, athletes who carry a lot of muscle mass would often be considered overweight if judged on BMI alone.
Obesity runs in families. Does that mean obesity is genetic or does it just mean that families share unhealthy eating habits? Perhaps surprisingly, twin studies have shown that obesity could be as much as 70% genetic, as siblings who share the same genes (identical twins) are much more likely to share the same body shape than non-identical twins (O’Rahilly & Farooqi, 2006).
Researchers have discovered a number of gene variants (alleles) that seem to be associated with obesity. Some of these are rare and affect a very small number of people, while others are fairly common and increase the risk of weight gain among large parts of the population (table 1).
| Rare single gene variants | Multiple common gene variants | |
|---|---|---|
| Effect on body weight | Account for a lot of extra weight in very few people | Account for a little bit of extra weight in a lot of people |
| Examples | Ob gene, Mc4r gene | Fto gene, Tmem18 gene |
| Association with other clinical conditions | Can be associated with rare diseases, e.g. congenital leptin deficiency, MC4R deficiency | One of many ‘normal’ varied human characteristics, but can also associate with other common diseases, e.g. type 2 diabetes |
| How are these found? | Candidate gene studies, animal studies, exome sequencing | Genome-wide association studies |
| Potential relevance | Prenatal genetic testing and gene therapy | Understanding risk of disease and tailoring disease prevention strategies |
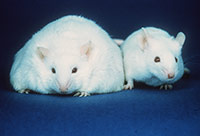
The Ob gene is a single-variant cause of obesity. It controls appetite by producing a hormone called leptin. People who carry two defective copies of this gene cannot produce leptin and have a constant desire for food. Obese, leptin-deficient people treated with leptin injections have returned to a normal weight. Another gene found to act in a similar way is the Mc4r gene, which is part of a signalling pathway that controls eating behaviour (O’Rahilly & Farooqi, 2006).
However, these single-gene mutations are quite rare. Some other, much more common gene variants have been identified that, although not a direct cause of obesity, do make carriers more likely to put on weight. Examples include variants of the Fto gene and the Tmem18 gene. Studies have found that people with one copy of the ‘at risk’ Fto variant weigh, on average, 1.2 kg more than people with other variants, whereas those with two copies of the ‘at risk’ variant weigh, on average, 3 kg more.
So are people who carry these more common ‘fat genes’ destined to be obese or can they escape their fate? These gene variants simply predispose people to gain weight, but with a healthy diet and lifestyle there is no reason why carriers shouldn’t be able to maintain a healthy weight.
Obesity researchers are also exploring epigenetics (the interactions between genes and environment) and how signalling pathways (e.g. hormones and the nervous system) in the body affect metabolism and behaviour. They hope that through a better understanding of the complex nature of appetite, metabolism and fat storage, it will be possible to develop better treatments or strategies for controlling food intake in people who are genetically predisposed to obesity.


The solutions can irritate eyes and skin, so wear a lab coat, safety glasses and gloves. Saliva can carry diseases; only handle your own loops or swabs and put used items in the disinfectant.
Disposal: liquids can be poured down the sink with plenty of water. Used loops or swabs can be placed in normal waste after disinfecting for 15 minutes.
This is just one of many published methods for extracting DNA from cells (e.g. Madden, 2006, and the Naked Scientists websitew2). They range from the very simple (using washing-up liquid and table salt) to methods that use chemicals more familiar to molecular biologists. The principles of the extraction are the same in all cases: a detergent is used to break up the cell membranes, an enzyme is added to digest the proteins that keep DNA tightly coiled up, and salt and cold alcohol are then added to create conditions where DNA is insoluble.
The main difference is that the more advanced the method, the purer the resulting DNA will be. For example, in the simplest method, a lot of pectin is mixed in with the DNA. Clearly, obesity researchers and other molecular biologists need their samples to be as pure as possible.
A ready-made sodium dodecyl sulphate (SDS) solution is recommended as powdered SDS is harmful if inhaled. If powdered SDS is used, the teacher should prepare the solution, wearing a mask and using the fume hood. See also the general Science in School safety note.
Suitable disinfectants include 0.015 M sodium hypochlorite solution, 1 % Virkon® solution or 5 % domestic bleach. After soaking for at least 15 min, the loops can be transferred to a plastic bag (wear gloves) and disposed of with normal waste.
To help you find the necessary reagents, a list of product numbers for Sigma-Aldrich is given in table 2. However, you will also be able to source them from other suppliers.
| Reagent | Sigma-Aldrich product number |
|---|---|
| Tris buffered saline (TBS) | Tablets: TS030 or 94158 |
| Sodium dodecyl sulphate (SDS) | Powdered: L3771 10% solution: 71736 |
| Sodium acetate | S2889 |
| Proteinase K | P6556 |
The incidence of obesity is increasing worldwide. Growing awareness of the problem, changing our eating habits and investigating a genetic predisposition could all contribute to its reduction.
This article, which explores recent research into the genetics of obesity and details a practical activity on the topic, could be used for biology lessons on many different topics, including health education, genetics, nutrition, pharmacogenomics, individualised therapy correlated with the presence of specific genes, statistics on obesity in different countries and their link to the diet of the corresponding population.
In addition to providing the starting point for a discussion of obesity, the article could be used for a project on nutrition, starting by recording students’ BMI and relating it to their daily diet (e.g. Krotscheck, 2010). In this way, the teacher could propose healthier eating habits, contributing to changing the attitude of students.
Suitable comprehension and extension questions include:
Panagiotis Stasinakis, 4th Lyceum of Zografou, Greece
Instructions for the activity (Word document)
Instructions for the activity (PDF file)
Download this article as a PDF